Gropman Laboratory
About the Lab
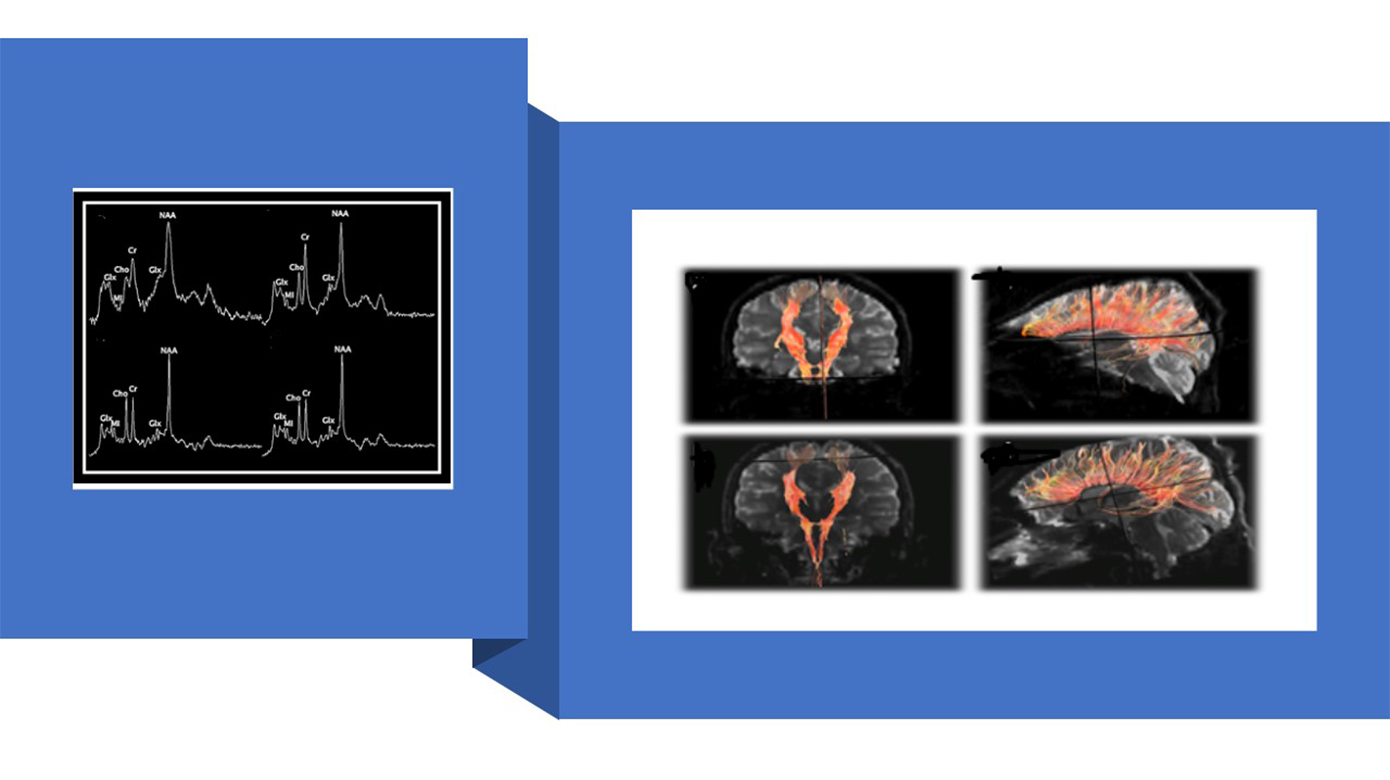
The Gropman Laboratory focuses on inborn errors of metabolism and chromosome disorders.
The two major categories of inborn errors studied are urea cycle disorders and mitochondrial disorders. The laboratory investigates brain biomarkers of injury using multimodal noninvasive technologies such as electroencephalogram (EEG), functional near infrared spectroscopy, magnetic resonance imaging (MRI) and magnetic resonance spectroscopy to investigate alterations in neural circuitry and metabolism in these disorders.
Andrea Gropman, M.D., is the principal investigator of the National Institutes of Health (NIH)-funded Urea Cycle Disorders Consortium (UCDC) and the site principal investigator of the North American Mitochondrial Disease Consortium (NAMDC). Both of these are part of the Rare Disease Clinical Research Network (RDCRN). Dr. Gropman oversees a Mitochondrial Center of Excellence at Children’s National Hospital. Her work in mitochondrial disorders also involves her role in the MitoEpiGen Program at George Washington University School of Medicine and Health Sciences, led by Anne Chiaramello, Ph.D. This project focuses on translational studies of promising therapeutics for patients with mitochondrial encephalopathy, lactic acidosis, stroke-like episodes (MELAS) and Leber hereditary optic neuropathy (LHON), two subcategories of mitochondrial disorders.
-
Lab Focus Areas
Brain injury, recovery and brain protection Intellectual and developmental disabilities
-
Partnerships
George Washington University School of Medicine and Health Sciences
Center for Functional and Molecular Imaging (CFMI)
Focus Foundation -
Contact
Andrea Gropman, M.D. Principal Investigator [email protected] 202-476-3511
Featured Publications
-
Neurodevelopmental outcome of prenatally diagnosed boys with 47,XXY (Klinefelter syndrome) and the potential influence of early hormonal therapy
Samango-Sprouse CA, Tran SL, Lasutschinkow PC, Sadeghin T, Powell S, Mitchell FL, Gropman A. PMID: 32220052 Am J Med Genet A 182(8):1881-1889. doi: 10.1002/ajmg.a.61561. August (2020) -
Molecular genetic and mitochondrial metabolic analyses confirm the suspected mitochondrial etiology in a pediatric patient with an atypical form of alternating hemiplegia of childhood
Gropman A, Uittenbogaard M, Brantner CA, Wang Y, Wong LJ, Chiaramello A PMID: 32489883; PMCID: PMC7262444 Mol Genet Metab Rep.28;24:100609. doi: 10.1016/j.ymgmr.2020.100609 May (2020) -
Hepatic arginase deficiency fosters dysmyelination during postnatal CNS development
Liu XB, Haney JR, Cantero G, Lambert JR, Otero-Garcia M, Truong B, Gropman A, Cobos I, Cederbaum SD, Lipshutz GS PMID: 31484827; PMCID: PMC6777909 JCI Insight 4(17):e130260 doi: 10.1172/jci.insight.130260 September (2019) -
The utility of EEG monitoring in neonates with hyperammonemia due to inborn errors of metabolism
Liu XB, Haney JR, Cantero G, Lambert JR, Otero-Garcia M, Truong B, Gropman A, Cobos I, Cederbaum SD, Lipshutz GS PMID: 30197275 Mol Genet Metab 125(3):235-240. doi: 10.1016/j.ymgme.2018.08.011. Epub November (2018) -
Investigating neurological deficits in carriers and affected patients with ornithine transcarbamylase deficiency
Sprouse C, King J, Helman G, Pacheco-Colón I, Shattuck K, Breeden A, Seltzer R, VanMeter JW, Gropman AL PMID: 24881970; PMCID: PMC4458385 Mol Genet Metab 113(1-2):136-41. doi: 10.1016/j.ymgme.2014.05.007 May (2014)
Related News
-

Research, Education & Innovation Week 2024
April 22, 2024 -

Your guide to Research, Education & Innovation Week 2024
April 15, 2024 -

Next-generation genomics testing holds key to undiagnosed rare disease
February 28, 2024
