Corbin Lab
Our Research
Uncovering the Developmental Genetic Programs for Establishment of Amygdala Neuronal Diversity and Connectivity
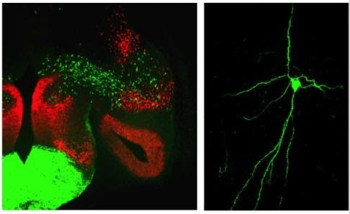
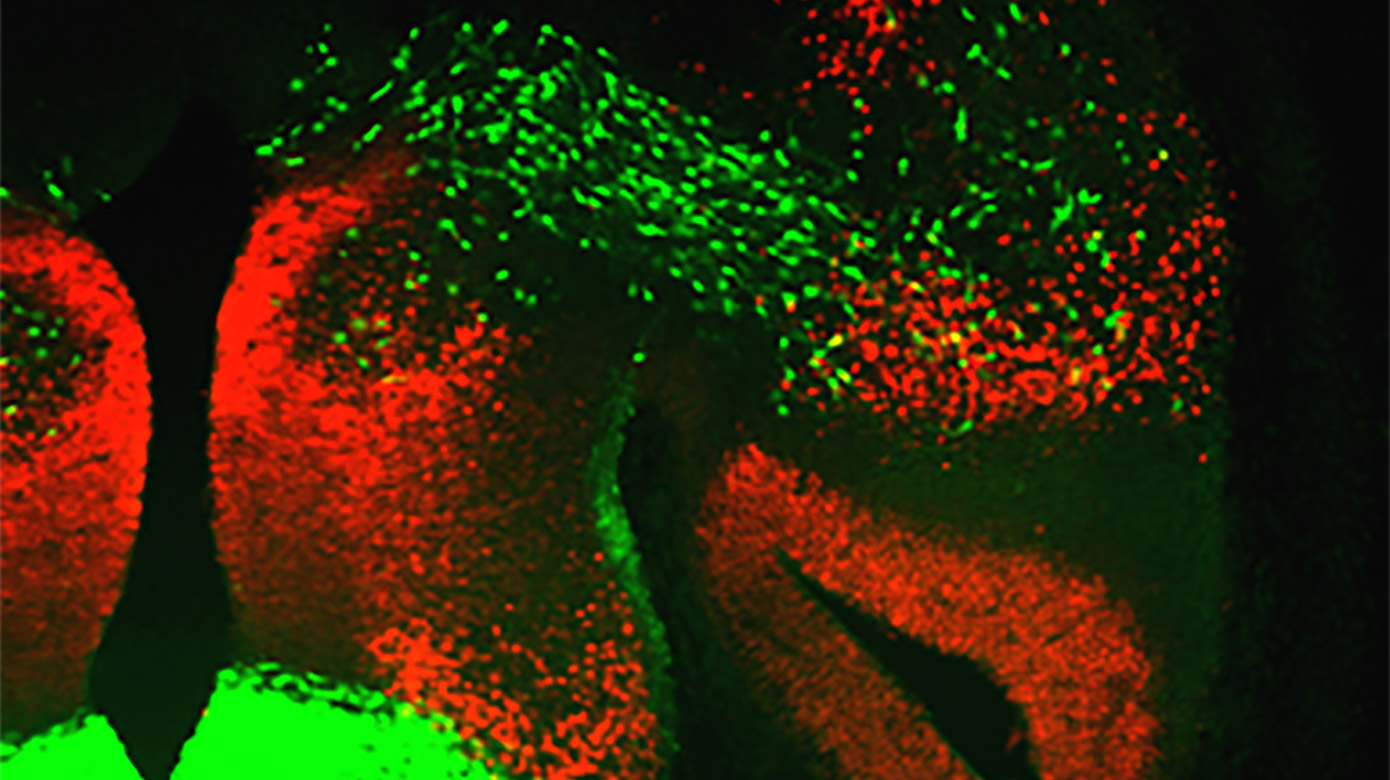
Proper displays of social interaction between members of the same species are required for species propagation and survival. These behaviors are regulated in large part via the amygdala, a central limbic system structure highly conserved across mammals. Alterations in specific aspects of amygdala development are a hallmark feature of human disorders such as autism and schizophrenia. While environmental inputs shape social behaviors, many components of these behaviors are hardwired. Thus, it is likely that developmental genetic programs pattern the wiring of brain circuits that control these behaviors. However, the genetic programs that drive formation of amygdala neuronal identity and circuitry remain largely unknown. We are currently addressing this question using a combination of developmental genetic, gene profiling, electrophysiological and behavioral approaches.Determination of How Developmentally Defined Limbic Subcircuits Control Sex-Specific Innate Social Behaviors
Despite a general understanding of how different brain structures control innate behaviors, the subcircuit logic for regulation of different innate behaviors remains generally unknown. Moreover, many of these behaviors differ between sexes suggesting different patterns of connectivity and/or circuit function in males and females.